Abstract
Background
Injection therapies are widely used for muscle injuries. As there is only limited evidence of their efficacy, physicians should be aware of the potential harmful effects of these injected preparations.
Objectives
The purpose of this review was to systematically review the literature on the myotoxic effects of intramuscular injection preparations commonly used for acute muscle injuries.
Data Sources
The databases of PubMed, Embase, Web of Science, Cochrane Library, CINAHL and SportDiscus were searched in March 2013.
Study Eligibility Criteria
Studies reporting histological evaluation or creatine kinase activity after intramuscular injection with local anaesthetics, corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs), platelet-rich plasma (PRP), Traumeel® and Actovegin®, or combination preparations were eligible for inclusion.
Data Analysis
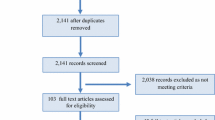
Two authors independently screened the search results and assessed the risk of bias. A best-evidence synthesis was used to identify the level of evidence.
Results
Forty-nine studies were included in this systematic review. There is strong to moderate evidence that intramuscularly injected local anaesthetics and NSAIDs are myotoxic, and there is conflicting evidence of the myotoxicity of PRP. There is limited evidence that single corticosteroid injections are not myotoxic but have a synergistic myotoxic effect when used together with local anaesthetics. There is no information to assess whether Actovegin® and Traumeel® are myotoxic.
Conclusion
Local anaesthetics and NSAID injections are not recommended for the treatment of muscle injuries in athletes, as they are myotoxic. The possible myotoxic effects of corticosteroids, PRP, Traumeel® and Actovegin® should be assessed in future research.

Similar content being viewed by others
References
Ekstrand J, Hägglund M, Waldén M. Epidemiology of muscle injuries in professional football (soccer). Am J Sports Med. 2011;39:1226–32.
Feeley BT, Kennelly S, Barnes RP, et al. Epidemiology of National Football League training camp injuries from 1998 to 2007. Am J Sports Med. 2008;36:1597–603.
Shankar PR, Fields SK, Collins CL, et al. Epidemiology of high school and collegiate football injuries in the United States, 2005–2006. Am J Sports Med. 2007;35:1295–303.
Orchard J, Seward H. Epidemiology of injuries in the Australian Football League, seasons 1997–2000. Br J Sports Med. 2002;36:39–44.
De Visser HM, Reijman M, Heijboer MP, et al. Risk factors of recurrent hamstring injuries: a systematic review. Br J Sports Med. 2012;46:124–30.
Levine WN, Bergfeld JA, Tessendorf W, et al. Intramuscular corticosteroid injection for hamstring injuries. A 13-year experience in the National Football League. Am J Sports Med. 2000;28:297–300.
Stevens KJ, Crain JM, Akizuki KH, et al. Imaging and ultrasound-guided steroid injection of internal oblique muscle strains in baseball pitchers. Am J Sports Med. 2010;38:581–5.
Ziltener J-L, Leal S, Fournier P-E. Non-steroidal anti-inflammatory drugs for athletes: an update. Ann Phys Rehabil Med. 2010;53(278–82):282–8.
Wright-Carpenter T, Klein P, Schäferhoff P, et al. Treatment of muscle injuries by local administration of autologous conditioned serum: a pilot study on sportsmen with muscle strains. Int J Sports Med. 2004;25:588–93.
Hamilton B, Knez W, Eirale C, et al. Platelet enriched plasma for acute muscle injury. Acta Orthop Belg. 2010;76:443–8.
Rettig AC, Meyer S, Bhadra AK. Platelet-rich plasma in addition to rehabilitation for acute hamstring injuries in NFL players clinical effects and time to return to play. Orthop J Sports Med. 2013;1(1). doi:10.1177/2325967113494354
Bernuzzi G, Petraglia F, Pedrini MF, et al. Use of platelet-rich plasma in the care of sports injuries: our experience with ultrasound-guided injection. Blood Transfus 2014;12 Suppl 1:S229–34.
Pfister A, Koller W. Treatment of fresh muscle injury. Sportverletz Sportschaden. 1990;4:41–4.
Lee P, Rattenberry A, Connelly S, et al. Our experience on Actovegin, is it cutting edge? Int J Sports Med. 2011;32:237–41.
Hamilton BH, Best TM. Platelet-enriched plasma and muscle strain injuries: challenges imposed by the burden of proof. Clin J Sport Med. 2011;21:31–6.
Sheth U, Simunovic N, Klein G, et al. Efficacy of autologous platelet-rich plasma use for orthopaedic indications: a meta-analysis. J Bone Joint Surg Am. 2012;94:298–307.
Orchard JW, Best TM, Mueller-Wohlfahrt H-W, et al. The early management of muscle strains in the elite athlete: best practice in a world with a limited evidence basis. Br J Sports Med. 2008;42:158–9.
Reurink G, Goudswaard GJ, Tol JL, et al. Therapeutic interventions for acute hamstring injuries: a systematic review. Br J Sports Med. 2012;46:103–9.
Rasmussen F, Svendsen O. Tissue damage and concentration at the injection site after intramuscular injection of chemotherapeutics and vehicles in pigs. Res Vet Sci. 1976;20:55–60.
Rasmussen F. Tissue damage at the injection site after intramuscular injection of drugs. Vet Sci Commun. 1978;2:173–82.
Zink W, Graf BM. Local anesthetic myotoxicity. Reg Anesth Pain Med. 2004;29:333–40.
Blom L, Rasmussen F. Tissue damage at the infection site after intramuscular injection of drugs in hens. Br Poult Sci. 1976;17:1–4.
Nosaka K, Sakamoto K. Changes in plasma enzyme activity after intramuscular injection of bupivacaine into the human biceps brachii. Acta Physiol Scand. 1999;167:259–65.
Diness V. Local tissue damage after intramuscular injections in rabbits and pigs: quantitation by determination of creatine kinase activity at injection sites. Acta Pharmacol Toxicol. 1985;56:410–5.
Nyska A, Skolnick M, Ziv G, et al. Correlation of injection site damage and serum creatine kinase activity in turkeys following intramuscular and subcutaneous administration of norfloxacin nicotinate. Avian Pathol. 1994;23:671–82.
De Morton NA. The PEDro scale is a valid measure of the methodological quality of clinical trials: a demographic study. Aust J Physiother. 2009;55:129–33.
Verhagen AP, de Vet HC, de Bie RA, et al. The Delphi list: a criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J Clin Epidemiol. 1998;51:1235–41.
Slavin RE. Best evidence synthesis: an intelligent alternative to meta-analysis. J Clin Epidemiol. 1995;48:9–18.
Van Tulder M, Furlan A, Bombardier C, et al. Editorial Board of the Cochrane Collaboration Back Review Group. Updated method guidelines for systematic reviews in the Cochrane Collaboration Back Review Group. Spine. 2003;28:1290–9.
Guttu RL, Page DG, Laskin DM. Delayed healing of muscle after injection of bupivicaine and steroid. Ann Dent. 1990;49:5–8.
Abe J, Fujii Y, Kuwamura Y, Hizawa K. Fiber type differentiation and myosin expression in regenerating rat muscles. Acta Pathol Jpn. 1987;37:1537–47.
Akiyama C, Kobayashi S, Nonaka I. Comparison of behavior in muscle fiber regeneration after bupivacaine hydrochloride- and acid anhydride-induced myonecrosis. Acta Neuropathol. 1992;83:584–9.
Basson MD, Carlson BM. Myotoxicity of single and repeated injections of mepivacaine (Carbocaine) in the rat. Anesth Analg. 1980;59:275–82.
Beitzel F, Gregorevic P, Ryall JG, et al. Beta2-adrenoceptor agonist fenoterol enhances functional repair of regenerating rat skeletal muscle after injury. J Appl Physiol. 1985;2004(96):1385–92.
Brazeau GA, Fung HL. An in vitro model to evaluate muscle damage following intramuscular injections. Pharm Res. 1989;6:167–70.
Brazeau G, Sauberan SL, Gatlin L, et al. Effect of particle size of parenteral suspensions on in vitro muscle damage. Pharm Dev Technol. 2011;16:591–8.
Carlson BM, Shepard B, Komorowski TE. A histological study of local anesthetic-induced muscle degeneration and regeneration in the monkey. J Orthop Res. 1990;8:485–94.
Cereda CMS, Tofoli GR, Maturana LG, et al. Local neurotoxicity and myotoxicity evaluation of cyclodextrin complexes of bupivacaine and ropivacaine. Anesth Analg. 2012;115:1234–41.
Chellman GJ, Faurot GF, Lollini LO, et al. Rat paw-lick/muscle irritation model for evaluating parenteral formulations for pain-on-injection and muscle damage. Fundam Appl Toxicol. 1990;15:697–709.
Cherng C-H, Wong C-S, Wu C-T, et al. Intramuscular bupivacaine injection dose-dependently increases glutamate release and muscle injury in rats. Acta Anaesthesiol Taiwanica. 2010;48:8–14.
Duguez S, Féasson L, Denis C, et al. Mitochondrial biogenesis during skeletal muscle regeneration. Am J Physiol Endocrinol Metab. 2002;282:E802–9.
Foster AH, Carlson BM. Myotoxicity of local anesthetics and regeneration of the damaged muscle fibers. Anesth Analg. 1980;59:727–36.
Fujikake T, Hart R, Nosaka K. Changes in B-mode ultrasound echo intensity following injection of bupivacaine hydrochloride to rat hind limb muscles in relation to histologic changes. Ultrasound Med Biol. 2009;35:687–96.
Grim M, Rerábková L, Carlson BM. A test for muscle lesions and their regeneration following intramuscular drug application. Toxicol Pathol. 1988;16:432–42.
Hall-Craggs EC. Rapid degeneration and regeneration of a whole skeletal muscle following treatment with bupivacaine (Marcain). Exp Neurol. 1974;43:349–58.
Horiguchi T, Shibata M-A, Ito Y, et al. Macrophage apoptosis in rat skeletal muscle treated with bupivacaine hydrochloride: possible role of MCP-1. Muscle Nerve. 2002;26:79–86.
Ishiura S, Nonaka I, Fujita T, et al. Effect of cycloheximide administration on bupivacaine-induced acute muscle degradation. J Biochem. 1983;94:1631–6.
Ishiura S, Nonaka I, Sugita H. Biochemical aspects of bupivacaine-induced acute muscle degradation. J Cell Sci. 1986;83:197–212.
Jiménez-Díaz F, Jimena I, Luque E, et al. Experimental muscle injury: correlation between ultrasound and histological findings. Muscle Nerve. 2012;45:705–12.
Jones GH. Protein synthesis in bupivacaine (marcaine)-treated, regenerating skeletal muscle. Muscle Nerve. 1982;5:281–90.
Lagrota-Candido J, Canella I, Pinheiro DF, et al. Characteristic pattern of skeletal muscle remodelling in different mouse strains. Int J Exp Pathol. 2010;91:522–9.
Louboutin JP, Fichter-Gagnepain V, Noireaud J. External calcium dependence of extensor digitorum longus muscle contractility during bupivacaine-induced regeneration. Muscle Nerve. 1996;19:994–1002.
McNeill Ingham SJ, de Castro Pochini A, de Oliveira DA, et al. Bupivacaine injection leads to muscle force reduction and histologic changes in a murine model. PMR. 2011;3:1106–9.
Nepomnyashchikh LM, Bakarev MA, Tsimmerman VG. Reparative reactions of the skeletal muscles in early aging OXYS rats with toxic metabolic injuries caused by bupivacaine. Bull Exp Biol Med. 2007;144:370–5.
Nonaka I, Takagi A, Ishiura S, et al. Pathophysiology of muscle fiber necrosis induced by bupivacaine hydrochloride (Marcaine). Acta Neuropathol. 1983;60:167–74.
Nosaka K. Changes in serum enzyme activities after injection of bupivacaine into rat tibialis anterior. J Appl Physiol. 1985;1996(81):876–84.
Nosaka K, Sakamoto K. Changes in plasma enzyme activity after intramuscular injection of bupivacaine into the human biceps brachii. Acta Physiol Scand. 1999;167:259–65.
Orimo S, Hiyamuta E, Arahata K, et al. Analysis of inflammatory cells and complement C3 in bupivacaine-induced myonecrosis. Muscle Nerve. 1991;14:515–20.
Osawa K, Maeda N, Kumegawa M, et al. Long-term observations of the masseter muscle following single or repeated injections of lidocaine hydrochloride into developing mice. Shika Kiso Igakkai Zasshi. 1989;31:198–210.
Plant DR, Colarossi FE, Lynch GS. Notexin causes greater myotoxic damage and slower functional repair in mouse skeletal muscles than bupivacaine. Muscle Nerve. 2006;34:577–85.
Politi PK, Havaki S, Manta P, et al. Bupivacaine-induced regeneration of rat soleus muscle: ultrastructural and immunohistochemical aspects. Ultrastruct Pathol. 2006;30:461–9.
Rosenblatt JD, Woods RI. Hypertrophy of rat extensor digitorum longus muscle injected with bupivacaine. A sequential histochemical, immunohistochemical, histological and morphometric study. J Anat. 1992;181(Pt 1):11–27.
Sadeh M, Czyewski K, Stern LZ. Chronic myopathy induced by repeated bupivacaine injections. J Neurol Sci. 1985;67:229–38.
Sadeh M, Stern LZ, Czyzewski K, et al. Alterations in creatine kinase, ornithine decarboxylase, and transglutaminase during muscle regeneration. Life Sci. 1984;34:483–8.
Sauerwein HP, Brouwer T, Dunning AJ. Creatine phosphokinase, myocardial infarction and intramuscular injection. Ned Tijdschr Geneeskd. 1975;119:1399–402.
Steiness E, Rasmussen F, Svendsen O, et al. A comparative study of serum creatine phosphokinase (CPK) activity in rabbits, pigs and humans after intramuscular injection of local damaging drugs. Acta Pharmacol Toxicol. 1978;42:357–64.
Tomas i Ferré J, Fenoll i Brunet R, Santafé M, et al. Changes in motor nerve terminals during bupivacaine-induced postsynaptic deprivation. J Anat. 1989;162:225–34.
White JP, Baltgalvis KA, Sato S, et al. Effect of nandrolone decanoate administration on recovery from bupivacaine-induced muscle injury. J Appl Physiol. 1985;2009(107):1420–30.
Yildiz K, Efesoy SN, Ozdamar S, et al. Myotoxic effects of levobupivacaine, bupivacaine and ropivacaine in a rat model. Clin Investig Med. 2011;34:E273.
Chellman GJ, Lollini LO, Dorr AE, et al. Comparison of ketorolac tromethamine with other injectable nonsteroidal anti-inflammatory drugs for pain-on-injection and muscle damage in the rat. Hum Exp Toxicol. 1994;13:111–7.
Ghozlan PR, Bernhardt M, Vélicitat P, et al. Tolerability of multiple administration of intramuscular meloxicam: a comparison with intramuscular piroxicam in patients with rheumatoid arthritis or osteoarthritis. Br J Rheumatol. 1996;35(Suppl 1):51–5.
Guterres SS, Fessi H, Barratt G, et al. Poly(rac-lactide) nanocapsules containing diclofenac: protection against muscular damage in rats. J Biomater Sci Polym Ed. 2000;11:1347–55.
Lima EM, Oliveira AG. Tissue tolerance of diclofenac sodium encapsulated in liposomes after intramuscular administration. Drug Dev Ind Pharm. 2002;28:673–80.
Narjes H, Türck D, Busch U, et al. Pharmacokinetics and tolerability of meloxicam after i.m. administration. Br J Clin Pharmacol. 1996;41:135–9.
Pyörälä S, Laurila T, Lehtonen S, Leppä S, Kaartinen L. Local tissue damage in cows after intramuscular administration of preparations containing phenylbutazone, flunixin, ketoprofen and metamizole. Acta Vet Scand. 1999;40:145–50.
Hammond JW, Hinton RY, Curl LA, et al. Use of autologous platelet-rich plasma to treat muscle strain injuries. Am J Sports Med. 2009;37:1135–42.
Harris NL, Huffer WE, von Stade E, et al. The effect of platelet-rich plasma on normal soft tissues in the rabbit. J Bone Joint Surg Am. 2012;94:786–93.
Wright-Carpenter T, Opolon P, Appell HJ, et al. Treatment of muscle injuries by local administration of autologous conditioned serum: animal experiments using a muscle contusion model. Int J Sports Med. 2004;25:582–7.
Campbell RSD, Dunn AJ. Radiological interventions for soft tissue injuries in sport. Br J Radiol. 2012;85:1186–93.
Rochcongar P, de Labareyre H, de Lecluse J, et al. L’utilisation et la prescription des corticoïdes en médecine du sport. Sci Sports. 2004;19:145–54.
Dietzel DP, Hedlund EC. Injections and return to play. Curr Pain Headache Rep. 2005;9:11–6.
Andia I, Sánchez M, Maffulli N. Platelet rich plasma therapies for sports muscle injuries: any evidence behind clinical practice? Expert Opin Biol Ther. 2011;11:509–18.
Titford M. The long history of hematoxylin. Biotech Histochem. 2005;80:73–8.
Miike T, Nonaka I, Ohtani Y, et al. Behavior of sarcotubular system formation in experimentally induced regeneration of muscle fibers. J Neurol Sci. 1984;65:193–200.
Komorowski TE, Shepard B, Okland S, et al. An electron microscopic study of local anesthetic-induced skeletal muscle fiber degeneration and regeneration in the monkey. J Orthop Res. 1990;8:495–503.
Seibel HR, Dolwick MF, Bush FM, et al. Electron-microscopic study of the rat masseter muscle following injection of lidocaine. Acta Anat. 1978;100:354–64.
Tamaki T, Akatsuka A, Uchiyama S, et al. Appearance of complex branched muscle fibers is associated with a shift to slow muscle characteristics. Acta Anat. 1997;159:108–13.
Mikkelsen UR, Langberg H, Helmark IC, et al. Local NSAID infusion inhibits satellite cell proliferation in human skeletal muscle after eccentric exercise. J Appl Physiol. 1985;2009(107):1600–11.
Hamilton B, Fowler P, Galloway H, et al. Nicolau syndrome in an athlete following intra-muscular diclofenac injection. Acta Orthop Belg. 2008;74:860–4.
Acknowledgments
No sources of funding were used in the preparation of this review. Gustaaf Reurink and Adam Weir reported receiving support from Arthrex Medizinsiche Instrumente GmbH for travel to a meeting about platelet-rich plasma. The other authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reurink, G., Goudswaard, G.J., Moen, M.H. et al. Myotoxicity of Injections for Acute Muscle Injuries: A Systematic Review. Sports Med 44, 943–956 (2014). https://doi.org/10.1007/s40279-014-0186-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-014-0186-6